Industrial wastewater refers to wastewater, sewage and waste liquid generated during industrial production, which usually contains industrial raw material residues, intermediate products, product by-products and various pollutants. With the acceleration of industrialization, the types and discharge of wastewater have increased significantly, leading to increasing water pollution and serious threats to the ecological environment and human health. Therefore, efficient treatment of industrial wastewater has become a key issue in environmental protection.

The application of TOC (total organic carbon) analyzers in industrial wastewater treatment has become particularly important. By detecting the concentration of organic carbon in water, TOC analyzers can effectively monitor the content of organic pollutants in wastewater. This technology can help quickly evaluate the effect of wastewater treatment and ensure that water quality meets discharge standards. Compared with traditional chemical oxygen demand (COD) analysis, TOC analyzers have higher sensitivity and accuracy, and are particularly suitable for industrial wastewater with complex components and multiple pollutants.
In industrial wastewater treatment, TOC analyzers can:
Real-time monitoring: Rapidly detect organic pollutants in wastewater, provide instant data, and help adjust the treatment process.
Optimize treatment processes: Help determine wastewater treatment needs by accurately measuring organic carbon content, ensuring efficient use of resources.
Improve treatment efficiency: Reduce the use of unnecessary chemical additives in the wastewater treatment process, reduce treatment costs, and improve the overall wastewater treatment effect.
TOC analyzers have gradually become a key tool in industrial wastewater treatment, providing more accurate and efficient wastewater monitoring solutions for various industries.

Next, we will share the experimental process of industrial wastewater testing using the WTOC3000 TOC analyzer.
Experimental part
Instrument and reagents
Instrument: Welso Total Organic Carbon (TOC) Analyzer (WTOC2000)
Reagents
Potassium hydrogen phthalate (reference reagent, used as TOC standard solution)
Sodium carbonate (reference reagent)
Sodium bicarbonate (superior purity)
Phosphoric acid (superior purity, used for acidification of samples)
Decarbonated distilled water (used for sample dilution and reagent preparation)
Experimental method
Preparation of standard solution
TOC standard solution (1000 mg/L)
Weigh 2.1254 g potassium hydrogen phthalate (pre-dried to constant weight at 110-120℃), dissolve in appropriate amount of pure water, transfer to a 1000 mL volumetric flask, dilute to scale with pure water, and mix well.
IC standard solution (1000 mg/L)
Weigh 4.4085 g anhydrous sodium carbonate (pre-dried to constant weight at 105℃) and 3.5000 g sodium bicarbonate (pre-dried in a desiccator), dissolve in appropriate amount of pure water, transfer to a 1000 mL volumetric flask, dilute to scale with pure water, and mix well.
Drawing of standard curve
Prepare mixed standard solution according to standard method, the concentration is as follows:
Total carbon (TC): 0.0, 4.0, 10.0, 20.0, 40.0, 80.0, 200.0 mg/L
Inorganic carbon (IC): 0.0, 2.0, 5.0, 10.0, 20.0, 40.0, 100.0 mg/L
Use the same volume and different concentration injection, measure the signal integral area, and draw the calibration curve with the mass concentration of carbon (mg/L) as the horizontal axis and the integral area signal as the vertical axis.
TC calibration curve equation:
Y=−1.643374×106X2+312868X+42Y=-1.643374\times10^6 X^2 + 312868X + 42Y=−1.643374×106X2+312868X+42
Correlation coefficient R=0.9999R = 0.9999R=0.9999
TIC calibration curve equation:
Y=−3.094839×106X2+338829X+20Y = -3.094839 \times 10^6 X^2 + 338829X + 20Y=−3.094839×106X2+338829X+20
Correlation coefficient R=0.9997R = 0.9997R=0.9997
Sample test
The sulfuric acid content in the two samples in this experiment is about 10%. In order to minimize the interference of sulfate ions on the test and to reduce the loss of instrument consumables, the samples are first manually injected and tested. 20μL of sample is drawn with a micro syringe and injected into the TC reaction tube. According to the test response value, the TOC content in the sample is between 400-600mg/L.
To ensure good test accuracy, the TOC concentration of the sample is diluted near the middle point of the standard curve, and the two samples are diluted 20 times with ultrapure water, and then the diluted samples are transferred to the sample bottle.
Test parameters
Cracking furnace temperature: 800℃
Carrier gas flow rate: 180 mL/min
Injection volume: 200 μL
Injection method: automatic injector
Test Results
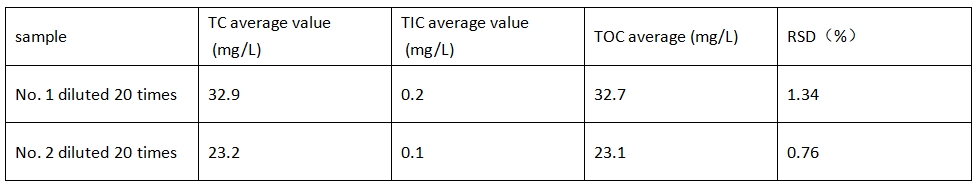
Multiplying the test results of the two samples by the dilution factor of 20, the TOC concentration of sample No. 1 was 654 mg/L, and the TOC concentration of sample No. 2 was 462 mg/L.

Results and Discussion
Common coexisting ions in samples such as SO₄²⁻, Cl⁻, NO₃⁻, PO₄³⁻, and S²⁻ may interfere with TOC testing. Carbon dioxide-free water is usually used to dilute the sample to reduce the concentration of coexisting ions to within the interference tolerance range before analysis.
When the sample TOC concentration is low, if the dilution factor is too large, the accuracy of the test results may be affected. Therefore, it is recommended to use the manual injection mode first, pre-determine the TOC concentration range, and then determine the appropriate dilution factor to ensure test accuracy.